Gemology
Physical Properties of Gemstones – Can we identify by body features?
Physical Properties of Gemstones

A gemstone’s optical properties can attract our eyes so it gives a cut and polished stone a unique character. However, the Physical Properties of Gemstones are also important. They determine a gem’s durability, guide gem cutters when they cut and set, and help us gemologists identify gemstones.
Specific Gravity – Physical Properties of Gemstones
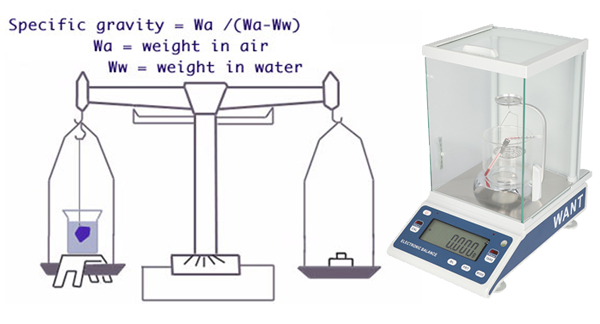 Specific gravity (SG) or density refers to how much something weighs in relation to its size. For example, a cubic centimeter of steel is much heavier than a cubic centimeter of styrofoam. SG is expressed as the relationship to an equivalent volume of water. For example, a gem with a specific gravity of 3 weighs three times as much as the same amount of water. In gemology, the terms specific gravity and density are interchangeable.
Specific gravity (SG) or density refers to how much something weighs in relation to its size. For example, a cubic centimeter of steel is much heavier than a cubic centimeter of styrofoam. SG is expressed as the relationship to an equivalent volume of water. For example, a gem with a specific gravity of 3 weighs three times as much as the same amount of water. In gemology, the terms specific gravity and density are interchangeable.
Specific Gravity Values of Popular Gemstones
Garnet 3.40-4.25
Corundum 3.97-4.03
Topaz 3.53-3.56
Diamond 3.51-3.53
Peridot 3.27-3.48
Tourmaline 2.84-3.10
Beryl 2.66-2.80
Quartz 2.63-2.68
Opal 1.99-2.25
Amber 1.05-1.096
As you see from the chart, few gems share a similar SG range. This makes SG one of every of the foremost useful gemstone physical properties you can have for identification. However, measuring specific gravity is difficult and time-consuming. Thus, you normally only conduct this test when necessary. (See “The Art and Science of Identifying Gemstones” for brand spanking new identification procedures that don’t typically require SG testing). Using specific gravity to Estimate Gem Weight When you’re buying and selling gems, knowing the SG of a gem can assist you to make the proper choice. For example, a one-carat opal with an SG of two would be much larger than a one-carat sapphire with an equivalent shape. Why? Because sapphire has an SG of 4. Let’s say you would like to seek out a replacement stone for a jewelry setting. While settings are measured in millimeters, gems are often sold by weight. If you order a one-carat sapphire to exchange a 1-carat opal, you’re certain an enormous surprise! If you recognize your gems well, you’ll estimate their weight by sight based on their SG.
Hardness and Toughness – Physical Properties of Gemstones The gemstone’s physical properties of hardness and toughness are often misunderstood. Their scientific meanings differ from their everyday meanings.
Hardness
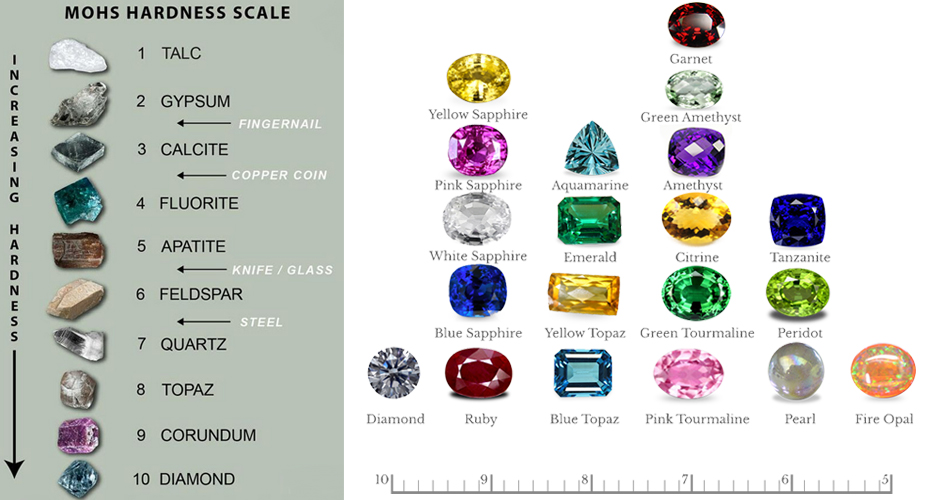
Scientifically, hardness is defined strictly because of the ability to resist scratching. Gemologists use the Moh’s Scale to define gemstone hardness. the scale runs from 1 to 10, with a selected mineral representing each number or unit. Diamond is the hardest at 10. Ruby comes in at 9, and so on. Each mineral on the scale will scratch softer stones, those below it. In turn, harder stones, those above them, will scratch it. Minerals of an equivalent hardness won’t scratch one another. 10 Diamond 9 Corundum 8 Topaz 7 Quartz 6 Feldspar 5 Apatite 4 Fluorite 3 Calcite 2 Gypsum 1 Talc
Moh’s Scale
Hardness has a very important effect on how well a gem will wear as jewelry. As a general rule (though exceptions abound), stones of hardness 7 or more will wear well. Since common dust consists largely of quartz (hardness 7), anything softer will eventually lose its polish. Even just wiping the dust off of a soft gem (below 7 in hardness) will cause fine scratches. Over time, these fine scratches accumulate and diminish the polish.
Toughness  Hardness has nothing to try and do with durability or toughness. Toughness or tenacity is defined because the ability to resist being hit. as an example, compare glass and wood. Scientifically, glass is far harder than wood. it’ll easily scratch the wood. However, you can hit a wooden board quite hard with a hammer without breaking it, yet glass will shatter with the slightest impact. So, while harder, glass isn’t nearly as tough as wood. You may not find toughness listed among gemstone physical properties in standard reference books. it’s no standard measurement procedures or units beyond verbal descriptions.
Hardness has nothing to try and do with durability or toughness. Toughness or tenacity is defined because the ability to resist being hit. as an example, compare glass and wood. Scientifically, glass is far harder than wood. it’ll easily scratch the wood. However, you can hit a wooden board quite hard with a hammer without breaking it, yet glass will shatter with the slightest impact. So, while harder, glass isn’t nearly as tough as wood. You may not find toughness listed among gemstone physical properties in standard reference books. it’s no standard measurement procedures or units beyond verbal descriptions.
Cleavage – Physical Properties of Gemstones 
Gemstone cleavage is a weak bond between the molecules of a mineral in certain planes in its crystal structure. you can best understand cleavage as a property of gemstones by comparing a gem to wood. Wood can split easily along the grain. However, splitting it across the grain is difficult. Some gems even have a direction along which they will easily split. this is often the cleavage plane. While cleavage affects toughness, it’s not related to hardness. Diamond is the hardest material known. However, since it’s cleavage planes, you’ll split it with a chunk of steel. Diamonds can break during normal wear. Cleavage fractures always run parallel to at least one plane of the first crystal’s surface. They’re always straight, flat, and difficult to examine if they’re inside the gem. you’ll spot them by the rainbow of colors that comes off a flat plane when light hits it at the right angle. Although hard to ascertain when small, cleavage fractures are vital for identifying and valuing a gem. Additionally, they indicate a weakness that would expand and ruin the gem.
Cleavage Descriptions
Gemologists describe cleavage in terms of how easily the material will part. the standard terms range from perfect, which separates very easily, to good, fair, and poor. you’ll come upon other terms, like imperfect. However, these are used less frequently. Another thing about descriptions is the number of cleavage planes. Some gems, like topaz, only have one direction of cleavage. Since topaz’s cleavage plane runs parallel to the bottom of the crystal, lapidaries usually cut it off at this axis. Thus, topaz is fairly durable despite its cleavage plane. Other stones, like feldspar, have cleavage on every crystal face. This makes cutting and setting problematic.
Please note that current cleavage terminology is a smaller amount than accurate. Take topaz, again. Because it’s described as having perfect cleavage, faceters realize it is often cut with minimum care. On the opposite hand, spodumene is an absolute monster to chop since it separates so easily. Still harder, diaspore comes during a class by itself. Yet, spodumene and diaspore are listed as having perfect cleavage, an equivalent to topaz and diamond. So, if you’re judging how well a stone will wear, look deeper into the cleavage than the easy listings.
Diamonds and topaz wear well. They’re commonly used as ring stones. However, kunzite, pink spodumene, and a few other gems would make very risky ring stones.
Fractures – Physical Properties of Gemstones 
Gemologists also describe how a mineral breaks in ways aside from along cleavage directions. They call this fracture. (Cleavage fractures are always flat, which makes them easy to spot and distinguish from other fractures). Although not always easy to call, fractures may offer you clues for gem identification. you’ll nearly always find fractures on the girdle or culet of a gem with a high-powered microscope. Of course, you’ll have better luck finding them on rough material. Fracture types include conchoidal, fibrous, splintery, granular, uneven, and hackly. The names are supported by what the fractures resemble. the foremost common sort of fracture is named “conchoidal”. You’ll see these on everything from glass to ruby.
Thus, they won’t help much with gem identification. However, discovering one among the less common fracture types would be a crucial clue. Keep in mind that fracture has nothing to try and do with the gemstone’s physical properties of hardness or toughness. Corundum is one among the foremost durable gems and it shares conchoidal fractures with glass, one among the foremost brittle. You can easily spot most of those fractures.
Conchoidal Typically, this fracture seems like an impact of a shell. If the fracture is a smaller amount complete, you’ll still see concentric banded lines during a curved section.
Fibrous Tiger’s eye gems have fibrous fractures.
Splintery Jade, ivory, and petrified wood (fossilized wood) often have splintery fractures. Be careful to not confuse fibrous and splintery. The difference is one among scale. Fibers are thin and fine, while splinters are usually thicker and coarser.
Granular fractures are common to lapis lazuli and maw-sit-sit.
Uneven Uneven fractures are often seen in sodalite and coral.
Hackly You’ll find hackly (sharp and jagged) fractures more commonly in metallic minerals.
Parting Like cleavage, parting occurs on a flat plane parallel to at least one of the crystal surfaces. Unlike cleavage, this results from gemstone twinning, where the crystal grew in overlapping layers. These layers have distinct thicknesses. Whereas a mineral with cleavage will have it in every specimen, parting only appears occasionally. It’s not present in every sample of a mineral species. for instance, you’ll find parts in some star rubies but not all of them. Star sapphires and rubies commonly contain parting. It’s somewhat common in chrysoberyl and quartz. You’ll usually see undercutting on the parting planes, as within the example pictured below. Undercutting represents a significant weakness during a gem and can greatly decrease its value and wearability.
 Stability – Physical Properties of Gemstones
Stability – Physical Properties of Gemstones
Stability refers to a gem’s ability to stay the same. Although most gems are very stable, don’t take that without any consideration. variety of things or sensitivities can affect the stability of certain gems. you ought to consider sensitivities generally, and light sensitivity especially, before you acquire a new gem, especially one you haven’t worked on. Leaving your beautiful, valuable, new acquisitions on your desk or during a bright case could cause them to change color or dry out.
Heat Sensitivity The most common gem stability problem is heat sensitivity. Gems don’t mind getting hot. Rather, the speed of change affects them. Thermal shock usually occurs during the cutting or setting of a gem. Lapidaries and metalsmiths need special training on the way to handle different gems to avoid thermal shock.
 Opals are so heat sensitive they even need special care when worn. you’ll cause them to crack by walking from a heated room into the winter cold or from an air-conditioned room into the complete summer sun. Since opals contain water, drying also can cause them to crack. To avoid cracking, store opals in an air-tight container or with a little piece of damp tissue. When wearing opals, keep them under your clothing if you’re going between environments with heat variations.
Opals are so heat sensitive they even need special care when worn. you’ll cause them to crack by walking from a heated room into the winter cold or from an air-conditioned room into the complete summer sun. Since opals contain water, drying also can cause them to crack. To avoid cracking, store opals in an air-tight container or with a little piece of damp tissue. When wearing opals, keep them under your clothing if you’re going between environments with heat variations.
Chemical Sensitivity  Certain chemicals can affect the look or composition of a gem with chemical sensitivity. This poses a serious problem for porous gems. for instance, turquoise and lapis lazuli can change color by absorbing oils from your skin. Therefore, don’t wear these gems while functioning on your car, painting, gardening, or doing anything where you can come in contact with chemicals. Certain acids can easily dissolve carbonates. These minerals include malachite, pearl, rhodochrosite, and marble. remember that a lot of common households and jewelry cleaners contain these acids.
Certain chemicals can affect the look or composition of a gem with chemical sensitivity. This poses a serious problem for porous gems. for instance, turquoise and lapis lazuli can change color by absorbing oils from your skin. Therefore, don’t wear these gems while functioning on your car, painting, gardening, or doing anything where you can come in contact with chemicals. Certain acids can easily dissolve carbonates. These minerals include malachite, pearl, rhodochrosite, and marble. remember that a lot of common households and jewelry cleaners contain these acids. 
Pearl is notoriously sensitive to chemicals. Always placed your pearl jewelry last when you’re getting dressed. Hairspray and atomized perfumes can ruin them.
Light Sensitivity 
The colors of some gems will fade if exposed to enough light. Such light-sensitive gems are called “evening stones,” since they’re best reserved for evening wear when they’re not exposed to sunlight. Even display case lights can damage light-sensitive gems. Thus, take care when displaying these gems for sale. (In addition, display lights can produce enough heat to dehydrate opals). Many gems suffer from light sensitivity to some degree. Some brown and gold topaz will fade slowly over time. As kunzite loses its color, it also loses a lot of value. Even within an equivalent species, sensitivity can vary. Blue spodumene, for instance, is somewhat light-sensitive. However, hiddenite (green spodumene) is so light-sensitive you ought to keep these rare specimens covered and only take them out for brief periods.
Artificially irradiated gems also can fade when exposed to light or heat.
Streak 
Optical properties largely determine a gem’s color. Streak, however, is one of the gemstone’s physical properties. It shows the color of a mineral without selective absorption live. To conduct a streak test, you powder a small little bit of a stone by rubbing the specimen across an unglazed porcelain tile or streak plate. Some stones have a streak color that differs considerably from what it shows under the light. for instance, most transparent colored stones, including emeralds, will show a colorless or white streak. Streak testing can help identify some stones. Hematite, an opaque metallic gemstone, features a reddish-brown streak, whereas hematite, a standard hematite imitation, features a brownish-black steak. However, don’t conduct this destructive test on finished stones. Test material in inconspicuous spots as a final resort only.
Magnetism Magnetism is one of the foremost misunderstood gemstone physical properties. The terms magnetism and magnetic refer to things possessing a magnetic field. Gemologists don’t use these terms to explain things that are attracted to magnets. Rather, they describe things that are magnets themselves. For instance, magnets attract ferrous metals. Hematite, a form of iron ore, would be attracted to a magnet. However, hematite itself isn’t magnetic, while hematine is. it’ll acquire metal objects.  Tests for identifying gemstone magnetism usually involve suspending the gem from a thread and holding it near a magnet. Although they work, you’ll put in a lot of labor for the small information you’ll receive. Keep this property in mind, but know there are other, much easier, and customary gem identification tests.
Tests for identifying gemstone magnetism usually involve suspending the gem from a thread and holding it near a magnet. Although they work, you’ll put in a lot of labor for the small information you’ll receive. Keep this property in mind, but know there are other, much easier, and customary gem identification tests.
Electrical Properties Minerals have a range of electrical properties.
Electroconductivity As the name implies, this is often the ability of a material to conduct electricity. Minerals with metallic bonding, like gold, silver, and copper, commonly possess this ability. a couple of minerals with partial metallic bonding are electrical semiconductors. Most gem minerals are nonconductors of electricity. However, there’s a crucial exception. Blue diamonds colored by artificial irradiation act as electrical insulators. Gemologists can distinguish them from naturally colored and synthetic blue diamonds by using thermal inertia meters.
Piezoelectricity  Some minerals, like colemanite, quartz, and tourmaline, generate electricity when placed under pressure. This property, piezoelectricity, is common in several minerals to varying degrees. When pressure is exerted at the ends, electricity will flow and make opposite positive and negative poles. due to this property, quartz and tourmaline have various industrial applications. for instance, thin slices of quartz can control frequencies for radios and watches. Some pressure gauges use tourmaline. Tourmaline was used to measure the blast pressure of the first atom bomb in 1945.
Some minerals, like colemanite, quartz, and tourmaline, generate electricity when placed under pressure. This property, piezoelectricity, is common in several minerals to varying degrees. When pressure is exerted at the ends, electricity will flow and make opposite positive and negative poles. due to this property, quartz and tourmaline have various industrial applications. for instance, thin slices of quartz can control frequencies for radios and watches. Some pressure gauges use tourmaline. Tourmaline was used to measure the blast pressure of the first atom bomb in 1945.
Pyroelectricity Some minerals generate an electrical current when heated. Examples include boracite, colemanite (also piezoelectric), rhodizite, and sphalerite.
Frictional Electricity  When rubbed, many gems commonly develop an electrostatic charge or frictional electricity. Notable examples include tourmaline and amber. If you rub them against something like wool, these gems gain enough charge to pick up ashes or small pieces of paper. the traditional Greeks recognized this quality in amber over 2,500 years ago. The word “electricity” comes from the Greek name for amber, Elektron.
When rubbed, many gems commonly develop an electrostatic charge or frictional electricity. Notable examples include tourmaline and amber. If you rub them against something like wool, these gems gain enough charge to pick up ashes or small pieces of paper. the traditional Greeks recognized this quality in amber over 2,500 years ago. The word “electricity” comes from the Greek name for amber, Elektron.
Thermal Conductivity  Some stones, notably crystalline gems, make excellent heat conductors. Because of their thermal conductivity, they’re going to draw heat far away from your fingers. Thus, they feel cool to the touch. Poor thermal conductors, like amber, glass, and plastic, feel warm to the touch. They don’t conduct heat far away from the body. If you concentrate on how a stone feels in your hand, you’ll often spot an imitation. When unsure, use a sensitive part of the body like your tongue or lips (but as long as you’ve recently cleaned the stone). Crystalline gemstone surfaces also will de-mist more rapidly than amorphous glass. Gemologists use thermal conductivity instruments to differentiate diamond, which conducts heat all right, from its simulants and imitations. Avoid drafts when testing, as they can change the readings. Thermal inertia meters measure how quickly the surface temperature of a gem changes when heated. This test also can help identify diamonds, since they need much higher thermal inertia than other gems.
Some stones, notably crystalline gems, make excellent heat conductors. Because of their thermal conductivity, they’re going to draw heat far away from your fingers. Thus, they feel cool to the touch. Poor thermal conductors, like amber, glass, and plastic, feel warm to the touch. They don’t conduct heat far away from the body. If you concentrate on how a stone feels in your hand, you’ll often spot an imitation. When unsure, use a sensitive part of the body like your tongue or lips (but as long as you’ve recently cleaned the stone). Crystalline gemstone surfaces also will de-mist more rapidly than amorphous glass. Gemologists use thermal conductivity instruments to differentiate diamond, which conducts heat all right, from its simulants and imitations. Avoid drafts when testing, as they can change the readings. Thermal inertia meters measure how quickly the surface temperature of a gem changes when heated. This test also can help identify diamonds, since they need much higher thermal inertia than other gems.
Hot Point Testing Thermal reaction testing, or hot point testing, also can distinguish real gems from imitations. You’ll use a pin heated over a flame to get smoke and odors from gem samples and show reactions to high heat, like melting or bubbling. Since melting is common to plastic, amber, wax, and resins, that result means your sample either is one among those materials or was surface treated with them. Odors can often be diagnostic. Tortoiseshell and a few corals emit a burning hair smell, while jet, fossilized coal, features a petroleum aroma. Amber produces a resinous smell and whitish smoke. Plastic imitations have an acrid, chemical odor. Odors also can reveal wax and plastic surface treatments applied to gems. For instance, turquoise should haven’t any smell. However, you’ll detect the odor of any treatment on the surface of the gem. Hot point testing is another destructive test. Test material in inconspicuous spots as the last resort only.
Physical Properties of Gemstones Article Composed by : සම්පත් සමරසේකර
Sampath Samarasekara
Chairman: Gemological Institute of Ceylon
Chairman: Youth Gem Professionals Association
Chairman: Sampath Gems
Chairman: Nanosoft Web Develop Company Direct WhatsApp: https://wa.me/message/2A4AUALYVQWRA1
Join with Gemological Institute of Ceylon for Practical Gemology Studies
Facebook Page: https://www.facebook.com/giceylon
Facebook Group: https://www.facebook.com/groups/1990653001208203
Facebook Profile: GI Ceylon
Websites: www.giceylon.com www.bluesapphire.lk www.gemluck.com
Join with Gem and Jewellery Business Help and Discussion Group on Telegram: https://t.me/joinchat/Q5cLnxilNw4xAr8T3TumfA
All images and text content are Copyrighted ©️


